So there’s little to no formal documentation from the perspective of an Intensivist on how to manage a CABG patient who is coming out of the CVOR to the CVICU. I’m sure some brilliant anesthesiologist or other intensivist has done this before, but I just haven’t found it. Let’s start. By the way, this is how I do it. This does not apply to every single patient. There are limitations, and there are complete textbooks written to include the nuances of how this is done for each and every scenario.
At the moment, this is a work in progress, and an algorithm for how I care for these patients will come next. I had an excellent colleague, Dr. Rishi Kumar, look at this for me to make sure I am not crazy. For a more comprehensive review of all things CVICU, CLICK HERE. None of this is medical advice, by the way.
Cite this post as: Eddy J. Gutierrez, “Recovering s/p CABG & Cardiac/Heart Surgery Patients in the CVICU”, eddyjoemd blog, November 22, 2021. Available at: http://eddyjoemd.com/cabg-cvicu/.
Nurses notify me that the are closing the CABG and getting ready to transport to the CVICU
This is when I start working on my note for the patient. I try to abstract as much data as I can about the patient from the electronic medical record (EMR). After all, when they arrive from the CVOR, I will not be able to obtain any history from them directly. After all, they will have an endotracheal tube in place. Here, I am looking for issues that may limit my ability to extubate them within 6 hours.
In addition, I am looking for home medications that may contribute to post-operative vasoplegia, such as ACE inhibitors or ARBs. Also, knowing if the patient is on an SSRI upfront will help you prepare in the rare case that a patient is severely vasoplegic. Methylene blue would be contraindicated on that patient. If the patient is chronically on glucocorticoids or has recently been on these medications, this can help precipitate adrenal insufficiency. Some anesthesiologists may use etomidate during induction. Remember that there’s a correlation between adrenal insufficiency and etomidate.
I also try to find an old but recent echocardiogram to have an idea of the starting point of the ejection fraction and the function of the right ventricle. Then, I compare it with what the cardiac anesthesiologist finds on his or her transesophageal echocardiogram (TEE). Then, I quickly build my consult or progress notes.
Direct Face to Face Handoff with the Cardiac Anesthesiologist
This step is paramount to understanding the patient’s physiology before and after their procedure. The optimal handoff is simple: they say “standard heart, nothing remarkable,” where, if anything, they are on the anti-hypertensive drip of choice at your shop. This is more of an exclusion rather than a rule. But let’s say it’s not a routine CABG; here is what I am asking or looking for the anesthesiologist to tell me, in no particular order.
- Airway: was it easy or difficult. If difficult, they’ll let you know what it took to secure it.
- The first numbers obtained when the swan-ganz catheter was placed (aka PA catheter) compared to the post-procedure numbers
- This is extremely important because I want to understand where the patient lived before their procedure.
- If a patient has elevated pulmonary artery pressures, like 70/40, before the procedure, I will not be too worried about 50/25 after the procedure. If the patient had pressures of 30/15 before the procedure but then 50/25 after the procedure, I will ask what happened. They can visualize the RV directly during the procedure and communicate its behavior.
- Someone who is used to living with a cardiac index of 1.5 may be able to escape with an index of 2.0 immediately post-op. If their index was 2.5 at first but is now 1.8, we’re missing something and it needs to be addressed.
- How much crystalloid was provide intra-op.
- Patients, from empirical knowledge, tend to receive somewhere between 1.5 and 3L in the OR. They may use the eyeball method using TEE to interpret how much fluids the patient should receive. Some use methods of fluid responsiveness, which I describe HERE. Remember that they cannot use heart-lung interaction methods, such as PPV, SVV, and EEO, since the chest is wide open. Knowing this number can help understand how much wiggle room one has to provide additional fluids before worrying about the heart’s actual function. I’ll talk about that later.
- How much the patient bled during the procedure.
- With all due respect to my surgeon friends, and this is a running joke in medicine, the documentation will say one thing, but the eyeballs and facial expressions of the anesthesiologist will say another. You will be told how much cell-saver and products were received and what was given to reverse the coagulopathy, and then one can sort out when to order repeat labs.
- Vasopressors or inotropes that are running and why.
- If the patient is on inotropes such as dobutamine or milrinone, they’ll tell you why they were started. For example, if a patient has a low cardiac index but also right ventricular dysfunction, some facilities will prefer milrinone. The different hospitals where I have practiced are inclined to choose dobutamine or milrinone for cardiogenic shock.
- Were the paralytics reversed on the patient?
- If they were, you can start working towards extubation. If not, depending on the agent used, you’ll know how long it’ll take for that dose to exit the patient’s system.
- What was the heart function?
- The anesthesiologist will offer information as to what the function of the heart was before and after the procedure. Here, one tends to fixate firmly on the ejection fraction, but knowing if the previously existing focal wall motion abnormalities have improved or resolved is also good information. There are places in the EMR where this can be looked up or even look up the images themselves, but hearing this directly is priceless. In valvular cases, one can get estimations of how the stenosis or regurgitation improved or resolved, and the collateral effects on the other valves are now experiencing quite a difference in pressure than they were accustomed to.
- The patients will come up on a temporary pacemaker. Dr. Rishi Kumar has a much better explanation than I could ever create, which you can access HERE.
- Let’s remember that they will tell you the procedure details.
- For example, which vessels were used if they did a 3-vessel CABG? Also, where they took the grafts from is essential. Don’t place a brachial arterial line on the same extremity where they just harvested a radial artery.
- Once the chit-chat has concluded, the anesthesiologist and their team return to the CVOR for their next case. Then, the nurses complete making their cables and tubes pretty (that’s a joke, by the way. Numerous messages have been received that nurses do more than make the cables look pretty. This is obvious. But they occasionally complain about the cables and lines being a mess, so I’m not changing the text I wrote.) The respiratory therapists obtain a baseline ABG (in some cases). We’re now in charge.
Hemodynamic management Immediately Post-Op
Two quick things: to understand how to optimize hemodynamics, we need to remember that MAP= (CO x SVR) + CVP. This is why the main numbers I pay attention to right off the bat are the cardiac index, the MAP, and the SVR. In addition, we need to remember that this is all a 6-hour recovery process for the majority of patients. The easy way to think of this is that the patients are in distributive shock and/or cardiogenic shock that tends to resolve on its own. We just need to support our patients until it does. We’ve got 6 hours to fix everything.
Analyzing the Cardiac Index
The rule of thumb number that I like to shoot for is 2.2L/min or higher. Here is why it’s important to know the baseline cardiac index as soon as the PA catheter insertion in the OR. If the patient has an index that is “saggy”, i.e. less than 2.2 or even less than 2.0, we need to investigate what has happened. This is where fluids MAY be helpful. The reason for this is that the whole point of providing fluids is to increase the preload and improve the stroke volume. Going back to the MAP equation listed above, MAP=(CO x SVR) + CVP, and CO = HR x SV. The whole point of giving fluids is to improve stroke volume. That is the definition of fluid responsiveness as explored thoroughly HERE.
Providing IV Fluids to Improve the Cardiac Index
You need to have a way to determine that the fluids, given via BOLUS and not by hitting 999ml/hr on the IV pump, is going to change stroke volume or cardiac index. I use approximately 250 or 500cc of lactated ringers. Albumin should not go in fast enough to change stroke volume. On the Vigilance II monitor that many of us have at our institution, we can see that the cardiac index is re-run periodically. The time interval can help us see if the index increases by 10% after a bolus of fluids. If the patient does not have a swan but rather a device that uses pulse-contour analysis, this device can be calibrated to calculate a delta stroke volume and determine whether they are in fact fluid responsive. If they are not fluid responsive, they DO NOT necessarily need more fluids nor would they benefit from them.
Don’t forget the heart rate. This is usually programmed into these patients via their external pacemaker but we need to be prepared for this to not be working properly. Is the heart appropriately contracting to the charges being provided by the pacemaker? If not, this needs to be corrected. If a temporary assistance is needed to get the index out of a bind, the HR can be increased on the pacemaker. This is NOT a definitive answer, though. This is to buy the patient time to sort out what is actually happening.
If the Cardiac Index is low and the the SVR is too high
Also take a quick look at the systemic vascular resistance. I will get into this a bit further but if the SVR is too high, i.e. 1500 where normal SVR is 800-1200, then the index is going to drop because there’s too much afterload on the heart. Some folks get a bit too enthusiastic with the norepinephrine when they notice that the patient is becoming hypotensive, then just add vasopressin, completely ignoring what the effects of those therapies are doing to clamp down the SVR and end up hurting the index. It makes the numbers look pretty until the patient crashes and burns. As an aside, this is something that happens in septic shock patients in whom the heart function is ignored. There is such a thing as septic cardiomyopathy and if one doesn’t identify this, then they are doomed to not do well.
s/p Fluids and the Cardiac Index is Still Low.
If the fluid status is appropriate and the SVR is just right, we need make sure that the patients hemoglobin is stable (i.e. not dumping a bunch of blood from their chest tubes, ensuring of course that these have been stripped appropriately by the nursing staff). If they need blood, provide them with blood.
Some inotropic assistance may temporarily be needed. Milrinone and dobutamine are often ordered with practice patterns depending on your institution. But do not forget that there may be something actually wrong with the heart. This is where performing a bedside echo will be helpful. Obtaining appropriate windows may be challenging because the patient just had their chest cracked open. Here, calling back the cardiac anesthesiologist to drop the transesophageal echocardiogram probe may be helpful.
Face to face handoff here helps you know who you should reach out to directly. They have photographic memories of what the heart looked like immediately after closure. If there are new focal wall motion abnormalities, time to call the surgeon. One of the grafts may have gone bad. If there is a growing pericardial effusion, strip the mediastinal tubes to see if this helps. But prepare to make that call to go back to the OR if the patient should go into cardiac tamponade.
Should the patient have had valvular surgery performed, checking the integrity of the valve may be needed here. A TEE would be in order and further management can be defined by this.
The Cardiac Index is abnormally high.
If, say, the cardiac index is 3.9, that also raises a red flag in my mind. This is where I take a look at the SVR because the patient might be vasoplegic and there’s no afterload on the heart to level things out. This is like you curling a 3lb dumbbell when you should be trying 20lb weights. It’s just too easy. If it’s too easy, this means something is wrong on the backend.
The Systemic Vascular Resistance
The way that I like to explain what happens when some patients go on pump is that they develop an allergic reaction, so to speak, and become vasoplegic. That’s definitely an oversimplification. Some go into distributive shock, which one of my partners, who is going to read this, LOVES when I document it in the chart. Vasoplegia is a type of distributive shock, for those who didn’t know. I remember the first time someone said vasoplegia to me in fellowship, I was confused as to what they meant.
Normal SVR is 800-1200 as mentioned above. Due to the inflammatory cytokines, interleukins and all that mumbo jumbo some patients aggressively vasodilate due to the circuit. We’ve gotten better at this over the years with the tubing and priming fluids, but we have not reached perfection yet.
So if the patient is hypotensive and the CI is fine, we need to look at the SVR. One isn’t able to directly visualize this on the Vigilance II monitor that most of us have if a PA catheter is in place, but it does populate on the “calc” section on your Philips monitor. If you have a device that uses pulse contour analysis via an arterial line, the SVR is displayed on the screen. If you want to calculate this by hand, good luck. SVR = (MAP – CVP) ÷ CO. There are also apps that will calculate this for you.
Add some vasopressors for hypotension + decreased SVR
If the patient is hypotensive and the SVR is low, this is the time to add vasopressors. My favorite to pull the trigger on here is usually norepinephrine. Now, if the patients PA pressures are too high, vasopressin may be a better choice. This is due to its lack of vasoconstrictive properties on the pulmonary arteries. See the images below for an example of what vasoplegia looks like.
Checking a blood gas, whether arterial or venous, can be helpful to identify if the patient is suffering an acidosis-induced vasoplegia. Is it respiratory nature? Adjust the vent. Is it metabolic? Some like to provide the bicarb-band aid here, i.e. give an amp of bicarb, but in my opinion sorting out the underlying etiology of the acidosis is key. There are opinions making arguments for and against the bicarbonate pushes that I will not go through here. If there is no acidosis, time to keep cruising through our differentials.
Do not arbitrarily give fluids
The knee-jerk reaction here is to provide additional fluids for said hypotension. Going back to our trusty equation, MAP = (CO x SVR) + CVP, one can see how giving fluids is not the answer. Constricting the dilated blood vessels is the answer. Remember, this vasoplegia is generally temporary and self-limiting. Having reviewed the home medications prior to getting here allows us to remember the likelihood that a home medication could be contributing to this vasoplegia.
Again, time is usually the answer here. But if it isn’t, this is where vasopressin is added, then possibly epinephrine. If the patient remains vasoplegic, methylene blue or cyanokit (if available) can be used. There’s also limited data for the use of angiotensin II (Giapreza) here. Stress-dose glucocorticoids are another option. Some studies are looking at IV Vitamin C for this indication due to ascorbic acid being a necessary co-factor in the production of endogenous catecholamines. Midodrine does not work here as the data does not support using midodrine in the CVICU as I discuss on THIS POST.
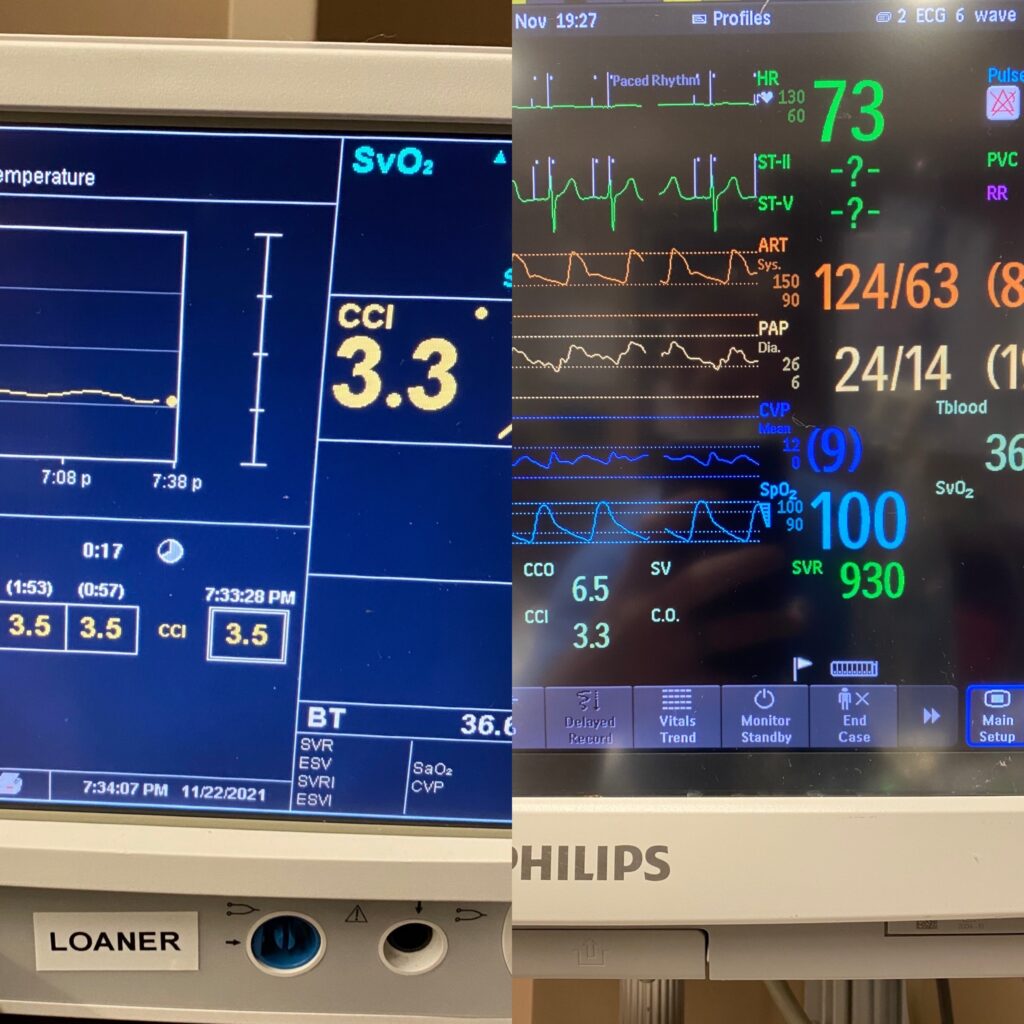
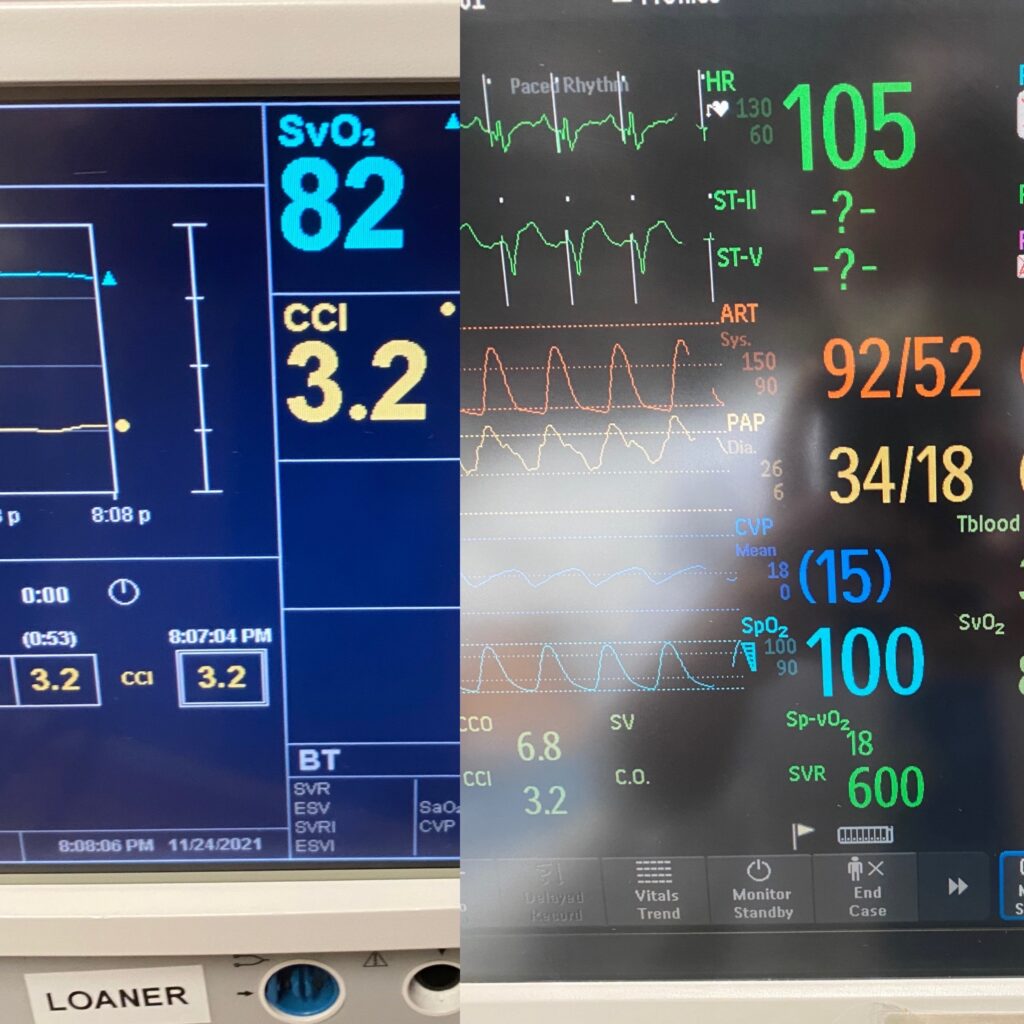
CVP: Not at the top of my priority list for CABG patients in the CVICU
Note that I have not discussed anything about the central venous pressure (CVP). I take the CVP with a grain of salt given all the known limitations of it since 1962. Beck’s triad doesn’t really work to diagnose pericardial effusion here because they all have muffled heart sounds after having a chest that has recently been cracked open. If the patient is hypotensive, they have a CVP that is equal to the PA diastolic pressure, with a subpar cardiac index, you need to make sure they aren’t going into tamponade. A bedside echo or a TEE should have an order in STAT. The patient may need to go visit the operating room again to relieve the pressure and stop the bleed causing this.
Understanding the Right Heart Function
Knowing what was going on with the right heart during surgery and immediately post surgery is helpful here. The likelihood that the RV will spontaneously go down is low but we need to think about it. If you want to use your cardiogenic shock data, you can calculate the pulmonary artery pulsatility index (PAPi). I describe PAPi superficially in THIS POST and in depth in THIS POST. I do not know if the PAPi score is valid for CVICU/CABG patients but it helps.
- PAPI=(systolic pulmonary artery pressure-diastolic pulmonary artery pressure)/CVP
- PAPI<0.9: indicates possible RV failure and that the clinician should consider RV support. Like everything in medicine, this is NOT an absolute.
- PAPI>0.9: indicates that the RV is likely normal. Some literature uses PAPI>1.0.
If your pulmonary artery pressures are too high, you may want to reach for a pulmonary vasodilator.
SvO2 in CABG Patients in the CVICU
I had written a full post about ScvO2 and SvO2 in the past that you can get your hands on HERE. Quickly touching up on my train of thought in these patients, the cardiac anesthesiologist could tell you what the baseline SvO2 was as soon as the PA catheter was floated. Then you’ll have an idea of where the patient lived before the procedure. You won’t be too concerned if the baseline was 40 and now after the CABG they’re sitting in the CVICU with an SvO2 is 56.
Remember that SvO2 has two components, O2 delivery (DO2) and consumption (VO2). In the delivery half of the deal, you should know that there are 3 parts to this: hemoglobin, cardiac output, and O2 consumption. To use SvO2 as a surrogate for cardiac output the other two need to be stable. In the VO2 side of things, you can’t use it reliably if your patient is tachypneic, febrile, shivering, or doing any nonsense that will contribute to increasing their O2 consumption. A rule of thumb for me is that over 60 is good enough. Over 70 is king. But every patient is different so the numbers here don’t rank as high on the importance list as others.
Mean Arterial Pressure
The mean arterial pressure NEEDS to be kept above 65mmHg in CABG patients in the CVICU. There’s data that even transient drops in blood pressure cause acute kidney injury. In addition, there are people who live in a hypertensive state. For them, even a MAP of 65 might not be high enough. Sorting this out is a fine balance. This is why patients in the PACU or in the CVICU have a nurse dedicated to them. Sometimes the need to have someone to help with the orders.
If a patient is bleeding, the surgeons may request to keep the MAP lower until it subsides or is under control. Keep in mind that you are robbing Peter to pay Paul with this strategy.
Bleeding of CABG patients in the CVICU
I cover this in more detail on THIS POST. Bottom line is that our nurses will notify us when the bleeding is too much. Our job is to provide blood products as needed. We make sure the coagulopathies are corrected. This includes providing calcium as needed due to the citrate causing hypocalcemia in the units of PRBCs. Our surgeon friends need to be notified sooner rather than later so that they can go back to the OR if needed. We need to call in a team and it’s best to do this while the sun is still out.
Extubation of CABG patients in the CVICU
Our goal is to pull the tube within the first 6 hours of them having their CABG. They leave the OR with the clock running the moment they hit the CVICU. If the patient is at least heading in the right direction, I will go ahead and extubate these patients. There’s good data to provide respiratory support with either the high-flow nasal cannula system or NIV should the patients have oxygenation or ventilation parameters that are on the cusp of normal. If the patient is on high doses of vasopressors, inotropes, is still bleeding, or has refractory issues, I will hold off on extubating them at that time. I guess that’s the main stuff. Let me know if there’s anything I may have missed.
Please let me know if there’s anything I missed regarding how an Intensivist should care for a CABG patient in the CVICU. Again, this is not medical advice nor is it all encompassing. Thanks again to Dr. Rishi Kumar for looking over this and doing a makeshift “peer review”. Grammar check by Jenny Moore, RN.
Consider purchasing my book, ‘The Vasopressor & Inotrope Handbook’!
I have written “The Vasopressor & Inotrope Handbook: A Practical Guide for Healthcare Professionals,” a must-read for anyone caring for critically ill patients (check out the reviews)! You have several options to get a physical copy. If you’re in the US, you can order A SIGNED & PERSONALIZED COPY for $29.99 or via AMAZON for $32.99 (for orders in or outside the US).
Ebook versions are available via AMAZON KINDLE for $9.99, APPLE BOOKS, and GOOGLE PLAY.
¡Excelentes noticias! Mi libro ha sido traducido al español y está disponible a traves de AMAZON. Las versiones electrónicas están disponibles para su compra for solo $9.99 en AMAZON KINDLE, APPLE BOOKS y GOOGLE PLAY.
When you use these affiliate links, I earn an additional commission at no extra cost to you, which is a great way to support my work.
Disclaimer
Although great care has been taken to ensure that the information in this post is accurate, eddyjoe, LLC shall not be held responsible or in any way liable for the continued accuracy of the information, or for any errors, omissions or inaccuracies, or for any consequences arising therefrom.

